Arrhythmogenic Right Ventricular Cardiomyopathy on the EKG
Arrhythmogenic right ventricular cardiomyopathy (ARVC), also known as arrhythmogenic right ventricular dysplasia, is a heart muscle disease clinically characterized by life-threatening ventricular arrhythmias and pathologically by an acquired and progressive dystrophy of the ventricular myocardium with fibrous or fibro-fatty replacement 1 2 3.
ARVC is an inherited condition which may lead to ventricular dysfunction, ventricular arrhythmias and sudden cardiac death. It is a major cause of sudden death in young individuals and athletes 1 2.
Epidemiology of ARVC
The estimated prevalence of arrhythmogenic right ventricular cardiomyopathy in the general population ranges from 1:2000 to 1:5000 and it affects more frequently males than females (up to 3:1) 2 3.
ARVC is reported as a cause of sudden cardiac death in 11%-27% of individuals aged ≤35 years 4.
Clinical Presentation of Arrhythmogenic Right Ventricular Cardiomyopathy
The initial phase is a preclinical phase, known as “concealed disease”, which is characterized by minimal or no structural abnormalities, with or without minor ventricular arrhythmias 2 5.
Arrhythmogenic right ventricular cardiomyopathy typically becomes clinically apparent between the second and fourth decades of life 2 3. More rarely, symptoms and signs can appear before puberty or in the elderly3.
The most common clinical presentation consists of ventricular arrhythmias and related symptoms or events, such as, palpitations, syncopal episodes (mostly occurring during physical exercise), and cardiac arrest 5.
Sudden cardiac death may be the first clinical manifestation of the disease, especially in young people and competitive athletes with a previously undiagnosed arrhythmogenic right ventricular cardiomyopathy 2 5.
ARVC may occasionally simulate an acute myocarditis, characterized by chest pain, transient ST-segment and T wave changes. Besides, a rise cardiac biomarker values may be detected.
Patients with long-standing disease may develop right ventricular or biventricular pump failure 2 6.
Diagnosis of Arrhythmogenic Right Ventricular Cardiomyopathy
At present, there are no gold-standard tests or pathognomonic criteria to make a definitive diagnosis of ARVC 1 3.
The diagnostic criteria, originally put forward in 1994 7, have been revised in 2010 to improve diagnostic sensitivity, but with the important prerequisite of maintaining diagnostic specificity 3.
The 2010 Task Force Criteria 8 facilitate diagnosis with an emphasis on major (each scores 2 points) and minor (each scores 1 point) criteria that include 6 domains 1 4 8:
- Structural abnormalities
- Histopathological findings
- Repolarization abnormalities
- Depolarization abnormalities
- Arrhythmia
- Family history
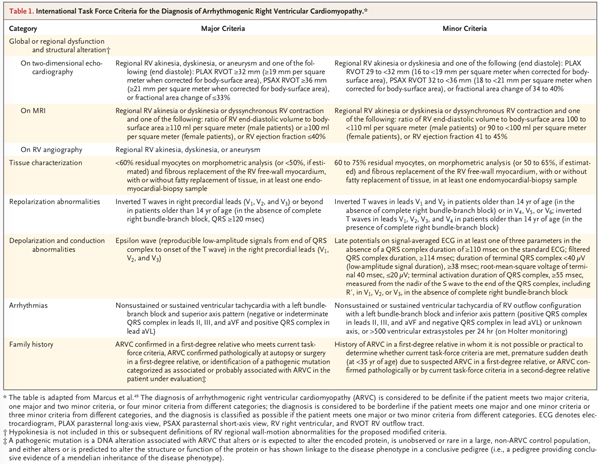
Criteria for arrhythmogenic right ventricular cardiomyopathy from Corrado D, Link MS, et al. Arrhythmogenic Right Ventricular Cardiomyopathy 2.
Each major criterion score 2 points, each minor criterion score 1 point, a diagnosis of ARVC requires 4 points in any of the following combinations: 2 major criteria, 1 major and 2 minor criteria, or 4 minor criteria 1 8.
The diagnosis is borderline if there are 3 points (1 major criterion and 1 minor criterion or 3 minor criteria) 1 8.
The diagnosis is possible if there are 2 points (1 major criterion, or 2 minor criteria) 1 8.
Although these criteria are more sensitive, they are difficult to robustly apply when used for screening family members and asymptomatic variant carriers and eliminating other possible diagnoses like myocarditis, sarcoidosis, dilated cardiomyopathy, Brugada syndrome, Uhl anomaly, Ebstein's anomaly, a left-to-right shunt, and pulmonary hypertension 1.
Electrocardiographic Findings on Arrhythmogenic Right Ventricular Cardiomyopathy
The electrocardiogram plays a key role in the diagnosis of ARVC 4.
Repolarization Abnormalities
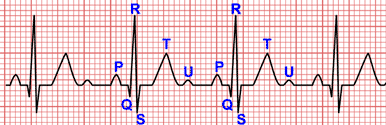
Repolarization abnormalities are early and sensitive markers of disease expression in ARVC 8. One of the key features of ARVC is the presence of T wave inversion in right precordial leads in patients over 14 years of age, in absence of complete right bundle branch block 4 8.
T wave inversion in leads V1, V2, V3, and beyond in healthy individuals >14 years of age is observed in only 4% of healthy women and 1% of men. Therefore, it is reasonably specific and considered a major diagnostic abnormality in ARVC 8.
Left ventricular involvement can occur or even be the main characteristic in ARVC. Negative T waves may extend to lateral precordial leads (V4, V5, or V6) suggesting left ventricular involvement 4 5.
Depolarisation abnormalities
Depolarization abnormalities include incomplete right bundle branch block (rarely complete), prolongation of right precordial QRS complex duration with a delayed S wave upstroke 5.
Epsilon Wave
Epsilon wave is a small amplitude potencial that occur at the end of the QRS complex and at the beginning of the ST-segment detected in the right precordial leads 4.
In ARVC, epsilon waves are thought to represent areas of delayed activation in the right ventricle as a consequence of fibrous and/or fibro-fatty replacement of right ventricular myocardium, for this reason epsilon waves are considered a major criterion 4.
Epsilon waves are recorded in patients with advanced disease 5.
Arrhythmias
Ventricular arrhythmias range from frequent premature ventricular complexes to ventricular tachycardia, which may degenerate into ventricular fibrillation. These arrhythmias are characteristically triggered or worsened by adrenergic stimulation 2.
The presence of more than 500 premature ventricular complexes in 24 hours is a minor criterion for the diagnosis of arrhythmogenic right ventricular cardiomyopathy 1 4 7.
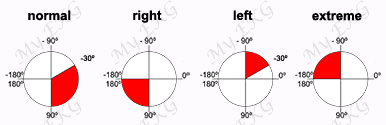
Not sustained or sustained ventricular tachycardias with a left bundle branch block and with superior axis (negative or indeterminate QRS in inferior leads, and positive in lead aVL) are considered a major criterion. Other patterns of ventricular tachycardia are considered a minor criteria 1 4 7.
Sustained ventricular arrhythmias may occurred in about 70% of the patients 6.
Electrocardiographic Criteria for Arrhythmogenic Right Ventricular Cardiomyopathy
Repolarisation abnormalities
Major
- Inverted T waves in right precordial leads (V1, V2, and V3) or beyond in individuals >14 years of age (in the absence of complete RBBB QRS ≥120 ms).
Minor
- Inverted T waves in leads V1 and V2 in individuals >14 years of age (in the absence of complete RBBB) or in V4, V5, or V6.
- Inverted T waves in leads V1, V2, V3, and V4 in individuals >14 years of age in the presence of complete RBBB.
Depolarisation/conduction abnormalities
Major
- Epsilon wave (reproducible low-amplitude signals between end of QRS complex to onset of the T wave) in the right precordial leads (V1 to V3).
Minor
- Late potentials by signal average EKG in one or more of 3 parameters in the absence of a QRS duration of ≥110 ms on the standard ECG:
- Filtered QRS duration ≥114 ms
- Duration of terminal QRS <40 μV (low-amplitude signal duration) ≥38 ms
- Root-mean-square voltage of terminal 40 ms <20 μV.
- Terminal activation duration of QRS complexs ≥55 ms measured from the nadir of the S wave to the end of the QRS complex, including R’, in leads V1, V2, or V3, in the absence of complete RBBB.
Arrhythmias
Major
- Nonsustained or sustained ventricular tachycardia of left bundle-branch morphology with superior axis (negative or indeterminate QRS in leads II, III, and aVF and positive in lead aVL).
Minor
- Nonsustained or sustained ventricular tachycardia of RVOT configuration, LBBB morphology with inferior axis (positive QRS in leads II, III, and aVF and negative in lead aVL) or of unknown axis, or more than 500 ventricular extrasystoles per 24h (on holter monitoring).
Treatment of ARVC
The most important goals of clinical management of patients with arrhythmogenic right ventricular cardiomyopathy comprise 3:
- Reduction of mortality, either by sudden death or due to heart failure.
- Prevention of disease progression leading to right ventricular, left ventricular or biventricular dysfunction.
- Attenuation of symptoms and improvement of quality of life by decreasing palpitations, ventricular tachycardia recurrences or ICD discharges.
- Reducing heart failure symptoms and increasing exercise capacity.
Therapeutic options consist of lifestyle changes, pharmacological treatment, catheter ablation, ICD mplantation, and heart transplantation.
The 2017 AHA/ACC/HRS Guidelines for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death 9 and the 2015 International Task Force Consensus Statement for Treatment of Arrhythmogenic Right Ventricular Cardiomyopathy/Dysplasia 10 recommend:
Lifestyle Changes
Patients with arrhythmogenic right ventricular cardiomyopathy have a significantly increased risk of sudden cardiac death during exertion 9 10. It is recommended that patients with a definite diagnosis of ARVD not participate in competitive and/or endurance sports and should avoid intensive exercise.
Pharmacological Therapy
Beta blockers are recommended in patients with ARVC and ventricular arrhythmias, to prevent effort-induced ventricular arrhythmias, and for the heart failure management 9 10.
Beta blockers can be useful in asymptomatic patients with ARVC and no ventricular arrhythmias or ventricular dysfunction 9.
Implantable defibrillator implantation
A prophylactic implantable defibrillator (ICD) implantation is also recommended in patients with high risk of sudden cardiac death (resuscitated sudden cardiac arrest, sustained ventricular tachycardia, significant ventricular dysfunction with RVEF or LVEF ≤35%) 9 10.
ICD implantation should also be considered in patients with ARVC and unexplained syncope , non sustained ventricular tachycardia, or moderate right or left ventricular dysfunction 9 10.
Catheter Ablation
Catheter ablation of ventricular tachycardias can be beneficial in patients with ARVC and recurrent symptomatic ventricular tachycardias in whom a beta blocker is ineffective or not tolerated 9 10.
Heart Transplantation
Patientes with ARVC and untreatable heart failure or uncontrollable ventricular tachyarrhythmias may require heart transplantation 10.
Heart transplantation is recommended as a final therapeutic option in ARVC patients with either severe, unresponsive congestive heart failure or recurrent episodes of VT/VF which are refractory to catheter ablation in experienced centres and/or ICD therapy 10.
References
- 1. Chahal A, Nosheen Reza N, Santangeli P. Risk Stratification in Arrhythmogenic RV Cardiomyopathy/Dysplasia Without an ICD. Expert Analysis. [Internet]. The American College of Cardiology; Feb 26, 2019 [cited: October 25, 2019]. Available on: https://www.acc.org/latest-in-cardiology/...
- 2. Corrado D, Link MS, Calkins H. Arrhythmogenic Right Ventricular Cardiomyopathy. N Engl J Med 2017; 376:61-72. doi: 10.1056/NEJMra1509267.
- 3. Pilichou K, Thiene G, Bauce B, et al. Arrhythmogenic cardiomyopathy. Orphanet J Rare Dis. 2016;11:33. doi: 10.1186/s13023-016-0407-1.
- 4. Quarta G, Elliott PM. Diagnostic Criteria for Arrhythmogenic Right Ventricular Cardiomyopathy. Rev Esp Cardiol. 2012; 65(7), 599–605. doi: 10.1016/j.rec.2012.02.015.
- 5. Corrado D, Basso C, Judge DP. Arrhythmogenic Cardiomyopathy. Circulation Research. 2017;121:784–802. doi: 10.1161/CIRCRESAHA.117.309345
- 6. Groeneweg JA, Bhonsale A, James CA et al. Clinical Presentation, Long-Term Follow-Up, and Outcomes of 1001 Arrhythmogenic Right Ventricular Dysplasia/Cardiomyopathy Patients and Family Members. Circulation: Cardiovascular Genetics. 2015;8:437–446. doi: 10.1161/CIRCGENETICS.114.001003.
- 7. McKenna WJ, Thiene G, Nava A, Fontaliran F, et al. Diagnosis of arrhythmogenic right ventricular dysplasia/cardiomyopathy. Task Force of the Working Group Myocardial and Pericardial Disease of the European Society of Cardiology and of the Scientific Council on Cardiomyopathies of the International Society and Federation of Cardiology. Br Heart J. 1994;71:215-218. doi: 10.1136/hrt.71.3.215.
- 8. Marcus FI1, McKenna WJ, Sherrill D, et al. Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: proposed modification of the Task Force Criteria. Eur Heart J. 2010 Apr;31(7):806-14. doi: 10.1093/eurheartj/ehq025.
- 9. Al-Khatib SM, Stevenson WG, Ackerman MJ, et al. 2017 AHA/ACC/HRS Guideline for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death. A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Circulation. 2018;138:e272–e391. doi: 10.1161/CIR.0000000000000549.
- 10. Corrado D, Wichter T, Link MS, et al. Treatment of Arrhythmogenic Right Ventricular Cardiomyopathy/Dysplasia. An International Task Force Consensus Statement. Circulation. 2015;132:441–453. doi: 10.1161/CIRCULATIONAHA.115.017944
If you Like it... Share it.